How evolving inspection expectations, digital manufacturing, and AI-enabled quality systems are reshaping pharmaceutical control strategies.
For many pharmaceutical organizations, the FDA’s draft guidance on Considerations for Complying with 21 CFR 211.110 may initially appear to be a routine clarification of sampling and testing expectations. Yet for those closely following the evolution of pharmaceutical manufacturing, the guidance represents something more significant, a light-bulb moment signaling how regulatory thinking is shifting. In many ways, the guidance signals an important shift in how regulators think about compliance from verifying quality through testing to demonstrating control through process understanding.
The message is clear: the future of pharmaceutical manufacturing will rely less on retrospective batch testing and more on real-time process understanding, digital monitoring, and data-driven control strategies. However, the successful adoption of these capabilities requires more than technological investment. It demands that organizations embed compliance, quality, and digital literacy into the culture of their operations. In this context, compliance becomes not merely a regulatory obligation but a strategic capability, one that enables organizations to adopt emerging technologies such as artificial intelligence while maintaining the highest standards of product quality and patient safety.
The evolution of pharmaceutical manufacturing control strategies reflects a broader regulatory shift from traditional batch verification toward real-time process understanding and proactive quality assurance as illustrated in Figure 1.
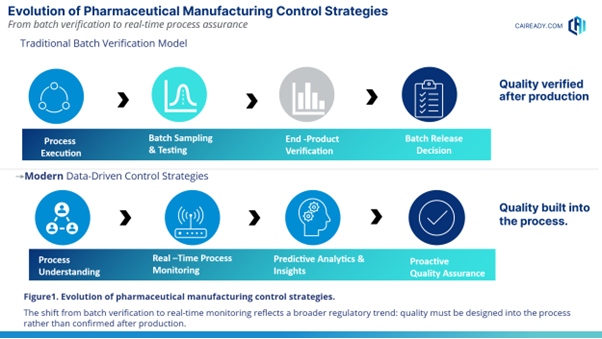
Understanding the Role of 21 CFR 211.110
Section 211.110 focuses on in-process controls, sampling, and testing during drug manufacturing. Its purpose is to ensure batch uniformity and product integrity through scientifically sound monitoring of production processes.
Historically, compliance with this regulation relied heavily on predefined sampling plans and laboratory testing throughout production. While effective, these approaches were largely retrospective, identifying issues only after samples had been analyzed. Today’s manufacturing environments are increasingly capable of generating continuous streams of process data. With advanced monitoring technologies and digital manufacturing systems, organizations can detect process variability earlier and respond proactively.The FDA’s draft guidance acknowledges this shift, clarifying how manufacturers can integrate advanced monitoring approaches while maintaining compliance with established GMP expectations.
FDA Inspection Trends: A Changing Regulatory Lens
The guidance also aligns with broader trends observed in recent FDA inspections. Regulators are increasingly evaluating the effectiveness of quality systems and process understanding, rather than focusing solely on isolated procedural deficiencies.
Inspection findings frequently highlight challenges related to:
- Insufficient process knowledge
- Ineffective CAPA systems
- Weak data governance
- Poor integration of digital systems within quality frameworks
This shift reflects a growing expectation that organizations demonstrate proactive control of manufacturing processes supported by reliable data and robust governance structures. In other words, inspection readiness is no longer an event; it is a continuous operational capability.
The Light-Bulb Moment for Pharmaceutical Manufacturing
The transformation underway can be understood through a simple shift in manufacturing philosophy from verifying quality after production to designing quality directly into the process. Achieving this shift requires more than technology, it demands integrated capabilities across culture, data infrastructure, and advanced analytics.
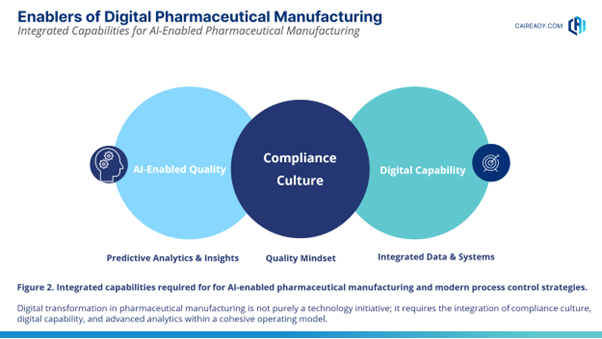
The Enablers of Modern Manufacturing
Three organizational capabilities are emerging as critical enablers of this transformation (Figure 2).
1. Compliance Culture
Quality and compliance must be embedded across the organization rather than residing solely within the quality unit. Employees across manufacturing, engineering, and digital teams must share accountability for maintaining product integrity and regulatory trust.
2. Digital Capability
Modern manufacturing environments depend on robust data governance, integrated digital systems (as illustrated in Figure 2), and workforces capable of interpreting complex datasets. Digital literacy is becoming a core competency for pharmaceutical professionals. Modern manufacturing environments depend not only on digital infrastructure but also on data literacy across the organization. As pharmaceutical operations increasingly generate large volumes of process data from manufacturing systems, sensors, and digital batch records, organizations must ensure that personnel understand how to interpret, contextualize, and act on these data streams. In practice, many companies are beginning to implement enterprise data platforms and data lake architectures that aggregate information from manufacturing execution systems, laboratory systems, and process monitoring tools. While these initiatives create powerful opportunities for advanced analytics and AI-driven insights, they also require strong data governance, standardized data models, and a workforce capable of translating raw data into meaningful process understanding and quality decisions. Without strong data literacy and governance, large-scale digital initiatives such as data lakes risk becoming repositories of information rather than engines for process insight and proactive quality management. Increasingly, regulators are also evaluating how organizations manage, interpret, and govern their manufacturing data during inspections, making data literacy and robust data governance essential components of modern quality systems.
3. AI-Enabled Quality
Artificial intelligence and advanced analytics offer powerful tools for identifying emerging process variability, predicting deviations, and strengthening decision-making within quality systems. However, these technologies must operate within well-governed frameworks that maintain transparency and regulatory confidence.
Questions Pharmaceutical Leaders Should Be Asking
For many pharmaceutical organizations, the question is no longer whether to adopt advanced manufacturing technologies, but how mature their implementation truly is. Leaders should consider several critical questions:
Are our advanced manufacturing systems inspection-ready?
Technologies such as continuous monitoring and digital process control must be integrated into formal control strategies and defensible during regulatory inspections.
Do we truly understand our processes or are we simply generating more data?
Data alone does not ensure control. Organizations must translate data into meaningful process understanding.
Is our workforce prepared for AI-supported quality systems?
Digital literacy and AI capability are becoming essential competencies across manufacturing and quality functions.
Do our quality systems support proactive risk detection?
Modern quality systems should identify emerging signals early rather than reacting to deviations after they occur.
Can we scale innovation without increasing compliance risk?
Implementing advanced technologies across multiple sites requires consistent governance, strong data management, and aligned regulatory strategies. As digital manufacturing systems become more sophisticated, an important question emerges: how will regulators evaluate AI-supported decision-making within pharmaceutical quality systems?
Looking Ahead: Toward Pharma 5.0
While the FDA’s draft guidance on 21 CFR 211.110 focuses on in-process controls and sampling strategies, it also reflects a broader shift in pharmaceutical manufacturing.
The industry is increasingly moving beyond digital transformation toward a more integrated model often described as Pharma 5.0, building on the foundations of Pharma 4.0.
Pharma 5.0 emphasizes the integration of advanced technologies, human expertise, and strong governance frameworks. In this model, AI and digital systems augment decision-making while a strong culture of quality ensures that innovation remains aligned with regulatory expectations and patient safety.
Conclusion
The FDA’s draft guidance on 21 CFR 211.110 may appear at first glance to be a technical clarification of in-process control requirements. Yet when viewed alongside evolving inspection trends and the increasing digitalization of pharmaceutical operations, it reflects a broader shift in regulatory expectations. The future of pharmaceutical manufacturing will depend not only on advanced technologies but on organizations that embed compliance culture, process understanding, and digital capability into their operational DNA.
Organizations that successfully align these elements will be best positioned to innovate with confidence, maintain regulatory trust, and deliver high-quality medicines in an increasingly complex manufacturing landscape. Ultimately, the organizations that succeed will be those that treat compliance not as a constraint on innovation, but as a foundation that enables it.
CAI supports pharmaceutical companies in navigating this transformation by strengthening quality systems, implementing modern manufacturing control strategies, and preparing organizations for increasingly digital and AI-enabled regulatory environments.
